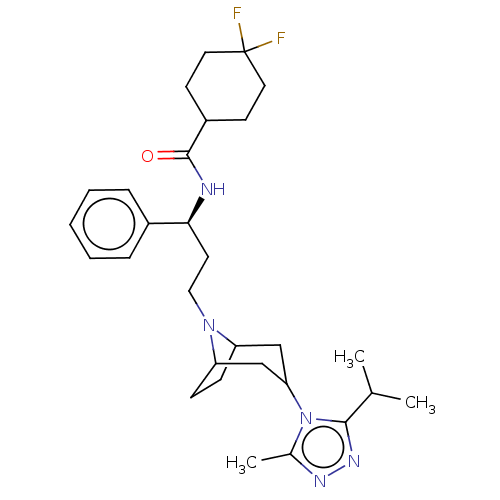
 BDBM160935 US9107954, maraviroc US10167299, Maraviroc
BDBM160935 US9107954, maraviroc US10167299, Maraviroc BDBM50334986 4,4-difluoro-N-((S)-3-(3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-aza-bicyclo[3.2.1]octan-8-yl)-1-phenylpropyl)cyclohexanecarboxamide 4,4-Difluoro-cyclohexanecarboxylic acid {(S)-3-[(1S,5S)-3-(3-isopropyl-5-methyl-[1,2,4]triazol-4-yl)-8-aza-bicyclo[3.2.1]oct-8-yl]-1-phenyl-propyl}-amide 4,4-difluoro-N-((S)-3-((1S,3S,5R)-3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-aza-bicyclo[3.2.1]octan-8-yl)-1-phenylpropyl)cyclohexanecarboxamide UK-427857 4,4-difluoro-N-((1S)-3-(3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-azabicyclo[3.2.1]octan-8-yl)-1-phenylpropyl)cyclohexanecarboxamide MARAVIROC Selzentry CHEMBL1201187
BDBM50334986 4,4-difluoro-N-((S)-3-(3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-aza-bicyclo[3.2.1]octan-8-yl)-1-phenylpropyl)cyclohexanecarboxamide 4,4-Difluoro-cyclohexanecarboxylic acid {(S)-3-[(1S,5S)-3-(3-isopropyl-5-methyl-[1,2,4]triazol-4-yl)-8-aza-bicyclo[3.2.1]oct-8-yl]-1-phenyl-propyl}-amide 4,4-difluoro-N-((S)-3-((1S,3S,5R)-3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-aza-bicyclo[3.2.1]octan-8-yl)-1-phenylpropyl)cyclohexanecarboxamide UK-427857 4,4-difluoro-N-((1S)-3-(3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-azabicyclo[3.2.1]octan-8-yl)-1-phenylpropyl)cyclohexanecarboxamide MARAVIROC Selzentry CHEMBL1201187
- Price, DA; Armour, D; de Groot, M; Leishman, D; Napier, C; Perros, M; Stammen, BL; Wood, A Overcoming HERG affinity in the discovery of the CCR5 antagonist maraviroc. Bioorg Med Chem Lett 16: 4633-7 (2006)
- Asano, S; Gavrilyuk, J; Burton, DR; Barbas, CF Preparation and activities of macromolecule conjugates of the CCR5 antagonist Maraviroc. ACS Med Chem Lett 5: 133-137 (2014)
- Lemoine, RC; Petersen, AC; Setti, L; Baldinger, T; Wanner, J; Jekle, A; Heilek, G; deRosier, A; Ji, C; Rotstein, DM Evaluation of a 3-amino-8-azabicyclo[3.2.1]octane replacement in the CCR5 antagonist maraviroc. Bioorg Med Chem Lett 20: 1674-6 (2010)
- Radioligand Binding Assay Afterwards, the pharmacological profile of bivalent ligand 1 at the chemokine receptor CCR5 was characterized similarly. The competitive radioligand binding assay was conducted in CCR5 rhesus macaque membrane preparations from Chem-1 cells. Monovalent ligand 3 and compound 4, an analogue of maraviroc, were tested along under the same condition. Introduction of the 4-NH2 group onto the phenyl ring of maraviroc, as seen in compound 4, caused approximately 65-fold decrease in the binding affinity, compared to maraviroc. The decrease of the binding affinity was even more profound for bivalent ligand 1 and monovalent ligand 3, as their Ki values dropped to submicromolar range, respectively.
- Radioligand Binding Assay A bivalent ligand 1 (FIG. 14) that combines the pharmacophores of naltrexone (a MOR antagonist) and maraviroc (a CCR5 antagonist) into one molecule was designed and synthesized. Herein is reported the characterization of this novel molecular probe in for its binding affinity, Ca2− flux functional activity, and HIV-1 inhibition potency. Bivalent ligand 1 was first characterized in hMOR-expressed CHO cells in the competitive radioligand binding assay as described previously.
- Flux Assay Then the Ca2+ functional activity of bivalent ligand 1 was evaluated in the Gqi5 transfected CCR5-MOLT-4 cells as described in the literature.3 As expected, no CCR5 agonism was detected for the bivalent ligand 1 (data not shown). In the RANTES induced Ca2+ flux inhibition assay (Table 3), the bivalent ligand 1 was approximately 60-fold less potent than maraviroc. A more significant potency decrease (nearly 300 times) was observed for the monovalent ligand 3, compared to maraviroc. In order to figure out the possible reasons for such a dramatic drop of their potency, two analogues (4 and 5, FIG. 14) of mavaviroc carrying gradient steric hindrance characters at the same substitution position were evaluated under the same condition. Compound 4 showed a modest reduction of the potency (Table 2). However, the inhibition potency of the N-t-Boc protected analogue 5 dropped to micromolar (IC50=1.57+/-0.18 uM). It thus appeared that steric hindrance may play an essential role.
 BDBM160935 US9107954, maraviroc US10167299, Maraviroc
BDBM160935 US9107954, maraviroc US10167299, Maraviroc BDBM50334986 4,4-difluoro-N-((S)-3-(3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-aza-bicyclo[3.2.1]octan-8-yl)-1-phenylpropyl)cyclohexanecarboxamide 4,4-Difluoro-cyclohexanecarboxylic acid {(S)-3-[(1S,5S)-3-(3-isopropyl-5-methyl-[1,2,4]triazol-4-yl)-8-aza-bicyclo[3.2.1]oct-8-yl]-1-phenyl-propyl}-amide 4,4-difluoro-N-((S)-3-((1S,3S,5R)-3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-aza-bicyclo[3.2.1]octan-8-yl)-1-phenylpropyl)cyclohexanecarboxamide UK-427857 4,4-difluoro-N-((1S)-3-(3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-azabicyclo[3.2.1]octan-8-yl)-1-phenylpropyl)cyclohexanecarboxamide MARAVIROC Selzentry CHEMBL1201187
BDBM50334986 4,4-difluoro-N-((S)-3-(3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-aza-bicyclo[3.2.1]octan-8-yl)-1-phenylpropyl)cyclohexanecarboxamide 4,4-Difluoro-cyclohexanecarboxylic acid {(S)-3-[(1S,5S)-3-(3-isopropyl-5-methyl-[1,2,4]triazol-4-yl)-8-aza-bicyclo[3.2.1]oct-8-yl]-1-phenyl-propyl}-amide 4,4-difluoro-N-((S)-3-((1S,3S,5R)-3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-aza-bicyclo[3.2.1]octan-8-yl)-1-phenylpropyl)cyclohexanecarboxamide UK-427857 4,4-difluoro-N-((1S)-3-(3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-azabicyclo[3.2.1]octan-8-yl)-1-phenylpropyl)cyclohexanecarboxamide MARAVIROC Selzentry CHEMBL1201187